AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Fission reaction equation examples1/9/2024  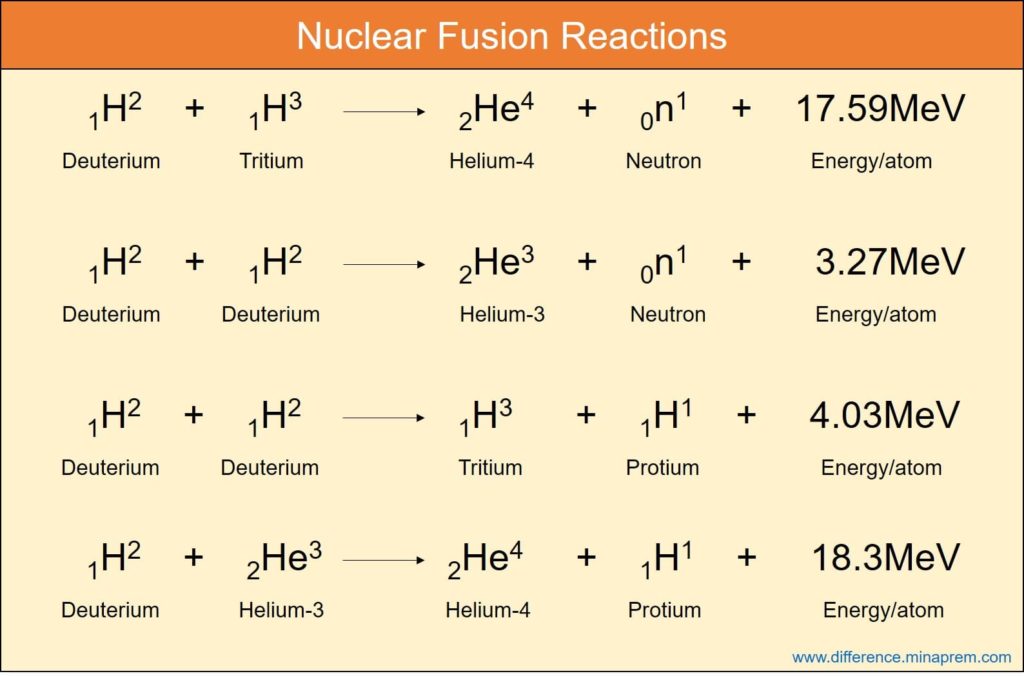
The fission of 1 kilogram of uranium-235, for example, produces about 2.5 million times as much energy as is produced by burning 1 kilogram of coal. “Such a reaction in which a heavy nucleus like that of uranium splits up into two nuclei of roughly equal size along with the emission of energy during the reaction is called fission reaction”.\)). Nuclear fission reactions produce incredibly large amounts of energy compared to chemical reactions. Secondly, a very large amount of energy is given out in this reaction.First, as a result of the breakage of the uranium nucleus, two nuclei of almost equal size are obtained, whereas in the other nuclear reactions the difference between the masses of the reactants and the products was not large.Uranium-235 is the main source of fuel for nuclear reactors. The fission reaction, which occurs in nuclear reactors, is the most well-known man-controlled nuclear reaction. Nuclear fission by radioactive decay is a natural process whereby a nucleus that is very big and unstable. Fission reactions generate large amounts of energy, which can be used to heat water to steam and power a turbine. The nuclear fusion reactions of light elements, which power the energy production of stars and the Sun, are among the most notable nuclear reactions. This process can happen naturally in the form of radioactive decay or in a nuclear reaction. This nuclear reaction was different from others to study other nuclear reactions, in two ways. Nuclear fission is the process of the nucleus of an atom splitting into multiple smaller parts and, in doing so, releasing a quantity of energy. A nuclear equation is an equation that describes the reactants and products in nuclear fission, nuclear fusion, or one of the four types of radioactive decay. It may be remembered that the mass of both krypton and barium is less than that of the mass of uranium. Hundreds of nuclear fission power plants around the world attest to the fact that controlled fission is practical and, at least in the short term, economical, as seen in Figure 32.20. Controlled fission is a reality, whereas controlled fusion is a hope for the future. When undergoing fission U-235 produces two medium-sized nuclei, and two or three neutrons. Nuclear fission is a reaction in which a nucleus is split (or fissured ). 
They observed that when slow-moving neutrons are bombarded on 23592U, then as a result of the nuclear reaction 14156Ba, 9236Kr, an average of three neutrons are obtained. Nuclear fission reactions produce incredibly large amounts of energy compared to chemical reactions. Otto Hahn and Fritz Strassmann of Germany while working on the nuclear reactions made a startling discovery. From above reaction it shows that 4 hydrogen nuclei. Nuclear Reactions: Fission 1) Write as much of the fission reaction as possible: 235/92 U + 1/0 n -> 97/40 Zr + 2(1/0 n) + 2) The atomic numbers must. 
In a nuclear fission reaction if we control the neutrons we can produce a fission chain reaction. Where QQ1+Q2+Q3, the total energy released during fusion of 4 hydrogen nuclei to form a helium nuclei. The fission process often produces gamma photons and releases a very large amount of energy even by the energetic standards of radioactive decay reactions. Example 10.6.1: Calculating Energy Released by Fission. Various products can be formed from this nuclear reaction, as described in the equations below. Nuclear fission is a reaction in which the nucleus of an atom splits into two or smaller nuclei. An important example of nuclear fission is the splitting of the uranium-235 nucleus when it is bombarded with neutrons.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
